Oldone asked
I already have the other shingles shot. Why now do they think I need a new type is shot?
Answer

Shingrix, the new shingles vaccine manufactured by GSK, is recommended by the CDC (Centers for Disease Control) for immunocompetent adults age 50 years and older without contraindications.
This recommendation stands even for individuals who have already received the Zostavax vaccine.
The reason for this recommendation is due to the fact that studies show Shingrix is more effective than Zostavax in preventing episodes of shingles. Furthermore, Shingrix is effective for a longer period of time.
Vaccine Efficacy
We discussed the efficacy of both Shingrix and Zostavax in another article:
The above article goes in-depth regarding efficacy rates, but to summarize:
- Shingrix: Herpes zoster infection rate per 1,000 patient-years = 0.3
- Shingrix: 97.2% overall efficacy rate
- Zostavax: Herpes zoster infection rate per 1,000 patient-years = 5.4
- Zostavax: 51% overall efficacy rate.
Additionally, the efficacy of the Zostavax vaccine decreases significantly with increasing age. The efficacy of Shingrix does not decrease to a significant extent in different age groups.
Below are the efficacy charts for both Zostavax and Shingrix (from their respective prescribing information inserts).
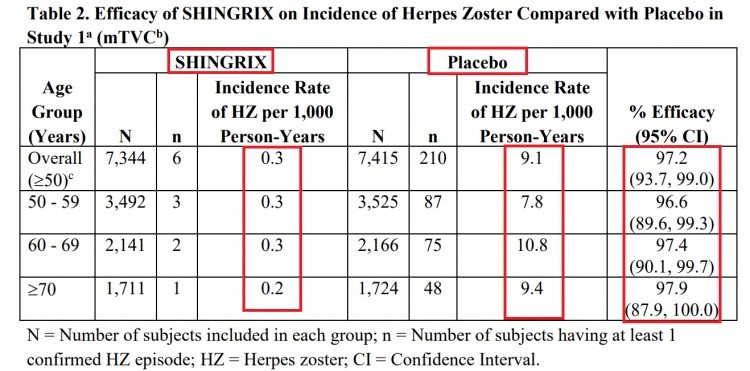
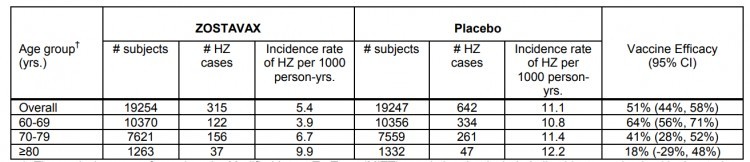
Shingrix Recommendations
The CDC recommendations for Shingrix in regard to Zostavax is as follows:
"CDC recommends two doses of Shingrix separated by 2 to 6 months for immunocompetent adults age 50 years and older:
- Whether or not they report a prior episode of herpes zoster
- Whether or not they report a prior dose of Zostavax"
Shingrix After Zostavax
The CDC gives more detailed information for those who have already received Zostavax:
"Consider the patient’s age and when he or she received Zostavax to determine when to vaccinate with Shingrix. Studies examined the safety of Shingrix vaccination five or more years after Zostavax vaccination. Shorter intervals were not studied, but there are no theoretical or data concerns to indicate that Shingrix would be less safe or effective if administered less than five years after a patient received Zostavax.
You may consider an interval shorter than five years between Zostavax and Shingrix based on the age at which the patient received Zostavax. Differences in efficacy between Shingrix and Zostavax are most pronounced among older patients. Studies have shown that the effectiveness of Zostavax wanes substantially over time, leaving recipients with reduced protection against herpes zoster."
Regarding the length of time to wait between Zostavax and Shingles, most sources in the United States recommend administering Shingrix as early as eight weeks after Zostavax.
In Canada, the recommendations are slightly different. They recommend waiting at least one year between Zostavax and Shingrix.
Be sure to discuss your vaccination options with your doctor.
Summary
- Shingrix is recommended for adults aged 50 and over, even if you have already been vaccinated with Zostavax.
- Recommendations for when Shingrix can be administered after Zostavax varies, but some sources state that vaccination with Shingrix can occur as early as 8 weeks after Zostavax (US Recommendation).
References
- Shingles (Herpes Zoster). CDC
- Zostavax Prescribing Information. AccessFDA
- Shingrix Prescribing Information. AccessFDA
- Shingrix Recommendations. CDC
- Herpes Zoster (Shingles) Vaccine: Canadian Immunization Guide. Canada Health
- Clinical Usage of the Adjuvanted Herpes Zoster Subunit Vaccine (HZ/su): Revaccination of Recipients of Live Attenuated Zoster Vaccine and Coadministration With a Seasonal Influenza Vaccine. PubMed




